The chemical reaction between zinc granules and dilute sulphuric acid is also characterised by a change in temperature (which is a rise in temperature). (iv) Change in temperature: The chemical reaction between quick lime water to form slaked lime is characterized by a change in temperature (which is a rise in temperature). There are some chemical reactions which can show more than one characteristics. (iii) Change in state of substance: The combustion reaction of candle wax is characterised by a change in state from solid to liquid and gas (because the wax is a solid, water formed by the combustion of wax is a liquid at room temperature whereas, carbon dioxide produced by the combustion of wax is a gas). The chemical reaction between sulphur dioxide gas and acidified potassium dichromate solution is characterized by a change in colour from orange to green. (ii) Change in Colour: The chemical reaction between citric acid and purple coloured potassium permanganate solution is characterised by a change in colour from purple to colourless. (i) Evolution of gas: The chemical reaction between zinc and dilute sulphuric acid is characterised by the evolution of hydrogen gas. 
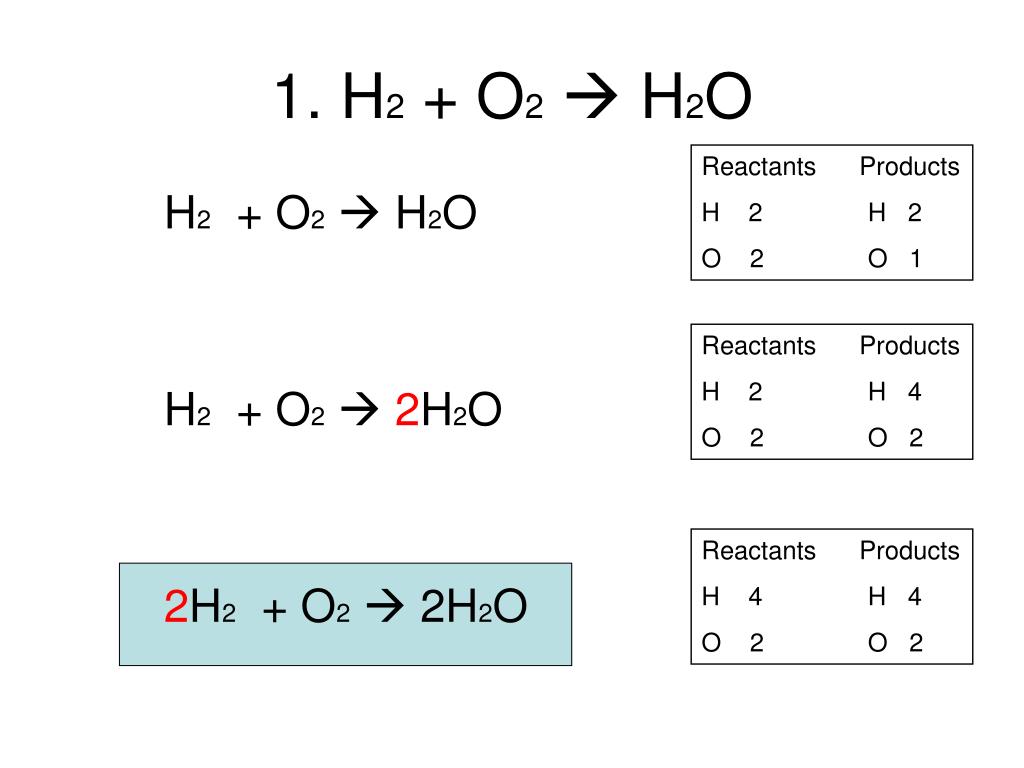
Product: New substance formed after a chemical reaction is called a product. Reactant: Substances which take part in a chemical reaction are called reactants. This is done to remove the protective layer of basic magnesium carbonate from the surface of the magnesium ribbon. The new substances produced as a result of a chemical reaction are called products.Įxample: The burning of magnesium in the air to form magnesium oxide is an example of a chemical reaction.ĢMg(s) O 2(g) \(\underrightarrow \) 2MgO(s)īefore burning in air, the magnesium ribbon is cleaned by rubbing with sandpaper.The substances which take part in a chemical reaction are called reactants.Only a rearrangement of atoms takes place in a chemical reaction. In a chemical reaction, a new substance is formed which is completely different in properties from the original substance, so in a chemical reaction, a chemical change takes place. For example: Rusting of iron, the setting of milk into curd, digestion of food, respiration, etc. What is a chemical reaction Class 10?Ĭhemical Reaction: The transformation of chemical substance into another chemical substance is known as Chemical Reaction. CBSE Class 10 Science Notes Chapter 1 Chemical Reactions and EquationsĬhemical Reactions and Equations: Balanced and unbalanced chemical equations and balancing of chemical equations. Here we have given NCERT Class 10 Science Notes Chapter 1 Chemical Reactions and Equations.Īccording to new CBSE Exam Pattern, MCQ Questions for Class 10 Science pdf Carries 20 Marks. Lakhmir Singh Science Class 8 SolutionsĬBSE Class 10 Science Notes Chapter 1 Chemical Reactions and Equations Pdf free download is part of Class 10 Science Notes for Quick Revision.PS Verma and VK Agarwal Biology Class 9 Solutions.NCERT Solutions for Class 9 Foundation of IT. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
